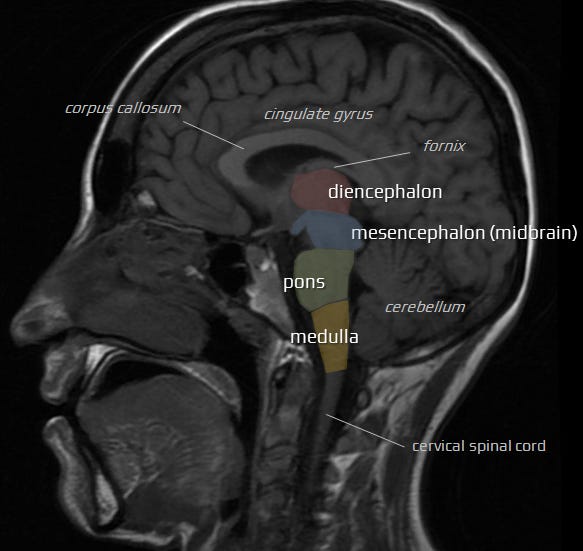
The role of the brainstem in ME/CFS and Long COVID - a minimal model.
Trying to connect disparate threads and findings into a coherent model of how acute infection can lead to long-term suffering.
ME/CFS is a highly multi-systemic disease, associated with disruptions to the cardiovascular system, the immune system, the nervous system, and the bio-energetic systems. Furthemore, these disruptions are severe, and impact fundamental functional and life-support systems in the body: the degree of disability that even moderate ME/CFS can produce is profound, and severe cases can be debilitating to a degree unmatched by almost any other disease; it is commonly cited that individuals with ME/CFS have a lower quality of life than even late-stage cancer patients. The breadth of systems impacted, and the severity of disability triggered, suggests that the fundamental etiology of ME/CFS probably will not lie in a single receptor, cell, organelle, or species of gut bacteria. Instead, I propose that it likely must involve a central regulator: some part of the human system able to impact almost every process in the body, including very fundamental ones such as sleep, immune activity, energy production, and sensory processing. The brainstem is a natural candidate for this, and as we will discuss below, several independent, converging lines of evidence suggest that the brainstem is fundamentally involved in the pathophysiology (and perhaps treatment) of ME/CFS. The brainstem is known to be involved in regulating almost all of the systems that are dysregulated in ME/CFS, including sleep/wakefulness, immune function, energy production, arousal, sympathetic and parasympathetic nervous system activity, and sensory gating.
In this post, I am going to propose a kind of minimal model of how brainstem damage could trigger symptoms of ME/CFS, how these can emerge following viral infection, and propose a small number of scientific studies that could be done to refine our understanding of brainstem function in ME/CFS in targeted ways. While this model is almost certainly an over-simplification, and does not claim to explain every symptom in every person, my hope is that by presenting a crystalized “toy model”, it will be possible to focus scientific and clinical attention in ways that help deepen our understanding of the true generative etiology of the disease.
I want to be very clear - none of these hypotheses or syntheses are unique to me. I am not claiming to have made some discovery or to have had some brilliant stroke of insight that never occurred to anyone else. Many of these ideas have, in various forms, been floating around online communities like Science 4 ME, Phoenix Rising, Health Rising, and others. I’ve consulted the excellent ME-Pedia reference a number of times, as well as writings by Jen Brea and Jeff Woods. My goal here is not to break new ground, but rather, to take all of these different ideas and crystalize them into a single, readable piece of accessible science which could be shared by patients or scientists.
Case studies brain-stem deformation as an etiology of ME/CFS
Full remission and recovery from ME/CFS is generally considered very rare, with meta-analyses suggesting that less than 5-10% of people achieve full recovery, and the majority of sufferers remaining afflicted for years or decades on end. In cases where total remission does occur, it is typically unclear why, or what change in the physiology produced such an unusual outcome. There are, however, two high-profile instances of people with severe ME/CFS who were able to determine the fundamental cause of their disease and correct it, thus achieving sustained remission through direct intervention. Both cases implicate the brainstem as a central locus of dysfunction in ME/CFS.
The first is the case of JW, who describes his experience on a personal website and has been active in the ME/CFS community. Following an acute infection of an unknown virus, JW developed debilitating ME/CFS with a classic presentation, including post-exertional malaise (PEM; the catastrophic worsening of symptoms following even modest exertion), profound fatigue, dysautonomia/POTS, and extreme sensory sensitivities, all of which left him bedbound for extended periods of time. Following a diagnosis of cranio-cerival instability (CCI) and atlantoaxial instability (AAI), JW discovered that vertical traction of his cervical spine caused a pronounced reduction in symptoms, including peripheral autonomic dysfunction. Following a successful skull-to-C2 spinal fusion, JW experienced sustained, total remission from ME/CFS.
The second case, JB, who has documented her experiences online as well, follows a remarkably similar trajectory: following an unknown viral infection (and possibly secondary to mold over-exposure), JB developed severe ME/CFS, with similar presentation to JW, including debilitating fatigue, dysautonomia/POTs, and PEM. Like JW, JB subsequently received a diagnosis of CCI, as well as tethered-cord syndrome, and discovered that vertical cervical traction dramatically relieved her symptoms. Following successful surgery for both spinal conditions, she achieved sustained remission from ME/CFS.
In both cases, the fundamental etiology of the ME/CFS was found to be compression of the brainstem: somehow, the viruses that JW and JB contracted triggered a loss of structural integrity in the cranio-cervical anatomy (coupled with pre-existing spinal deformation in the case of JB) that resulted in the weight of the skull and brain essentially squishing the brainstem. This squishing prompted a cascade of subsequent energetic, sensory, and immunological effects that manifested as severe ME/CFS. Once this squishing was corrected via traction or surgical intervention, normal brainstem function was (apparently) restored and both reported rapid and sustained remission from their years-long, progressive illness.
Since these initial, high-profile case studies, there has been an effort among the community of people with ME to track signs and symptoms of cranio-cervical dysfunction, and among those who ultimately do receive neurosurgery to correct it, what the outcomes are. A survey from the Phoenix Rising forum has recorded a large number of respondents with both ME/CFS and a variety of diagnosed spinal issues, as well as a number who went into remission following surgery. There have also been a very small number of published case studies of improvement from chronic fatigue syndrome following spinal surgery (three are presented in the Journal of Translational Medicine).
In the interest of presenting a full picture and minimizing unnecessary risk, I want to stress that the kinds of invasive spinal surgeries that JB and JW got are not panaceas and should be approached with caution. While there have been some remarkable success stories of remission following surgery, there have also been a number of failures. The same Phoenix Rising record shows plenty of cases of individuals who did not have these near-miraculous remissions, and some even worsened. A friendly acquaintance of mine from college with ME/CFS got the procedure done and remains severely ill; if you have some extra money, you can donate to his GoFundMe.
What do we make of these various anecdotes and case studies? Clearly there are some people for whom spinal surgery is remarkably effective, and others for whom it is at best neutral or at worst actively damaging. The surgery itself is also a brutal affair and even in successful cases leaves people with significantly restricted mobility in their neck - it would be nightmare to go through such a terrible ordeal only to find yourself still severely ill with ME/CFS and now unable to move your neck. Is there anything we can learn from these successes and failures that will help us refine a hypothesis about the brainstem and the different ways it can be involved (and which treatments might or might not help)?
ME/CFS, CCI, and IIH - The mechanical basis hypothesis
Five (admittedly remarkable) case studies of remission from severe ME/CFS is absolutely not grounds to claim that every single person with the condition has a mechanical basis for their disorder - the cases of surgical failure reinforce this idea. However, it does present what mathematicians might call an existence proof. It seems undeniable that deformation or other stress to the brainstem can cause prototypical ME/CFS symptoms (although it does not logically follow that all, or even most, prototypical ME/CFS symptoms are caused by mechanical brainstem stress).
A natural next question might be: how common is this? Are JB, JW, and the small number of respondents gathered on the Phoenix Rising poll outliers? Strange cases of convergent symptoms arising from an unusual and atypical etiology? It’s currently impossible to say, since there hasn’t been much formal research on what JW calls the “mechanical basis hypothesis”, and nothing even remotely resembling a clinical trial. However, there is a current of research exploring a closely-related issue in ME/CFS: idiopathic intracranial hypertension (IIH).
Intracranial hypertension occurs when the pressure of cerebral-spinal fluid (CSF) inside the skull is too high (either because too much is produced, or too little can drain out), and it can physically deform the structure of brain tissue, including the brainstem. In a large study of 205 volunteers with ME/CFS, 55% were found to have indicators in IIH (as evidenced by inflated optic-nerve sheath diameter) and 83% signs of possible IIH. The same study found that, of 125 volunteers who received MRIs, 80% had some kind of obstruction to the cervical spine analogous to what JB had. An earlier study of 20 patients with ME/CFS who received lumbar punctures, 25% were found to have diagnosable IIH, and 85% (including all five of the IIH individuals) reported symptomatic relief associated with cerebral-spinal fluid drainage. The same group also reported a case study of a woman who had suffered from ME/CFS for many years and was found to have borderline IIH. She reported “life-changing” remission following the placement of bilateral venous stents that allowed the CSF to drain normally. Collectively, these results suggest that a sizable proportion of people with ME/CFS have a mechanical disruption to the basal brain, either in the form of CCI, IIH, or both.
As of yet, there hasn’t been any clinical trials attempting to treat IIH (or other cranio-cervical issues) to ameliorate ME/CFS symptoms. While the bar for surgeries like spinal fusion or implanting venous stents is very high (and won’t get through ethics review boards without a much more robust base of preclinical evidence), there is an interesting, much less invasive test: Diamox. Diamox is a medication that lowers intracranial pressure and is currently prescribed for diagnosed cases of IIH. According to a Health Rising article, at least some physicians treating ME/CFS patients have added it to their arsenal of treatments, and anecdotal reports on communities like /r/cfs suggest that it can make a significant difference to some people. Given that Diamox is a much much less invasive intervention than spinal surgery, with much less risk of catastrophic worsening, I would personally encourage any interested person with ME/CFS to consider the IIH angle prior to considering the surgery angle (although I am not a doctor, and this not medical advice in any way, shape, or form).
Given ME/CFS is typically understood as a multisystem disease implicating everything from the immune system to the gut, the discovery that brainstem deformation and pathology appears to be a widely conserved feature of ME/CFS necessitates asking “are there any mechanisms by which localized damage could produce such distributed dysfunction?”
The answer increasingly appears to be “yes.”
Brainstem-based immune regulation
I’ve been thinking about the brainstem in ME/CFS for a while, ever since finding JW and JB’s stories online, but the idea that this could be important really began to crystalize after seeing a recent paper in Nature, which showed compelling evidence that a particular population of neurons in the brainstem mediates immune response via the vagus nerve. The paper itself is very technical, but from the ten-thousand foot view, the story is quite simple: using mice as an animal model, Jin et. al, found that activating the immune system with an injection of a noxious substance triggered activity in part of the brainstem known as the caudal nucleus of the solitary tract (cNST). The cNST is an important mediator between the immune system, the autonomic nervous system, and the central nervous system; it is a major part of the vagus nerve complex, and also projects to the thalamus and the hypothalamus (which are involved in sensory processing and hormonal function respectively - both of which can become deranged in severe ME/CFS). Where it gets very interesting (and why I suspect this paper got into Nature) is that the researchers were able to test the causality going the other way. By using some biological wizardry, they were able to “silence” the cNST, essentially turning it “off”, which allowed them to see how the central nucleus regulated the peripheral immune system.
The result of silencing the neurons was “a run-away, out-of-control inflammatory response;” pro-inflammatory cytokines are mass-produced (an increase of almost 300%, while anti-inflammatory cytokines are suppressed. This effect also runs in reverse: stimulating activity in cNST causes a global calming of immune activity, suppression of pro-inflammatory cytokines, and an increase in anti-inflammatory cytokines (an almost ten-fold increase, in fact). It appears that the cNST serves as a regulator, constantly suppressing immune activity by sending signals down the vagus nerve to the peripheral nervous system, and on into the immune system.
Inflammation is thought to be a key part of the pathophysiology of ME/CFS, with different inflammatory markers correlating with the severity of a variety of symptoms. Neuroinflammation in particular may be of special import and has been found to be widespread throughout the brain in PET studies as well as in post-mortem tissue samples. It’s even right there in the name “encephalomyelitis”, which means “inflammation of the brain and spinal cord.” Similarly, the involvement of the vagus nerve in regulation of the immune system by the cNTS is also significant, as vagal and parasympathetic dysfunction is a typical feature of ME/CFS and long COVID, including during sleep.
In the study, silencing the cNST was achieved with complex, and thoroughly unnatural manipulations (none of which I understand). However, a natural question might be: could the cNST also be silenced by “squishing” the brainstem? Either with the weight of the skull or pressure from excessive CSF pressure? Unfortunately, the paper cannot answer this question, but I was able to find a fascinating literature on immune system dysregulation following traumatic brain or spinal cord injury. Chronic inflammation following spinal cord injury is well-documented, and while none of the papers I found mention the cNST specifically, there is plenty of evidence of immune derangement following brain and spinal cord injury.
Other symptoms generally associated with ME/CFS can occur following traumatic brain injury, such as POTS-like symptoms appearing after concussion, extreme fatigue, and sleep disturbances. The overall similarities between post-concussion syndromes and ME/CFS are profound enough that some researchers are looking into TBI research as a way to gain insights into post-viral ME/CFS and long COVID.
The elephant in the room, then, is the question of what any of this has to do with viruses. While a minority of people with ME/CFS may develop it after physical trauma, the vast majority of sufferers do so following an infection (typically with a virus, although bacterial infections such as Lyme disease can do so as well). This was the case for both JW and JB, the two survivors whose ME/CFS were found to have a root cause of brainstem compression. How could a virus cause traumatic brain injury?
The connective tissue link
I suspect that the answer lies in connective tissue dysfunction. It has been long recognized that connective tissue disorders, particular hypermobile Ehler-Danlos syndrome (hEDS), is a particular risk factor for developing ME/CFS. One study put the rates of diagnosable hEDS among people with ME/CFS at 20%, and hEDS severity predicted ME/CFS symptom burden. These findings were replicated in another study, that found 15% of people with ME/CFS qualified for joint hypermobility, which was associated with more severe symptom presentation as well. Hypermobility has also been found to be a risk for long COVID. Beyond the direct overlap between connective tissue disorders and ME/CFS, it is also known that hEDS is associated with many of the issues we linked to ME/CFS above. People with hEDS are at risk of idiopathic, intracranial hypertension, cranio-cervical instability, and Mast cell activation syndrome. The evidence is overwhelming that dysfunctional connective tissue both increases individual risk for post-viral illness, as well as many of the associated syndromes that accompany it.
Not everyone with ME/CFS or long COVID is hypermobile, however, and many people who do have both post-viral and connective tissue disorders were able to live normal lives prior to the virus that triggered their ME/CFS, even with defective connective tissue. The infection (or perhaps the body’s response to infection) is the inciting incident. Something changes in the body after the infection, something that people with connective tissue problems are at greater risk of, but could conceivably strike anyone. The fact that connective tissue problems are a risk suggests that the natural place to look would be at how infections alter the structural integrity of the body’s connective tissue: after all, those with hEDS and related disorders may be vulnerable because their bodies already lack structural integrity and cannot afford to lose what they have.
Is it possible that a viral infection, even a mild one, could permanently damage connective tissue? This is an area where I cannot find a tremendous amount of research, although what I’ve found certainly suggests that it might be possible. A recent population-level study of over a million people from Korea found that COVID-19 can induce connective tissue disorders and a systematic review of the literature also found the same. Both of these studies looked particularly at autoimmune connective tissue disorders, but I also found plenty of evidence that viral or bacterial infection can also acutely trigger degradation of connective tissue. Part of the normal immune response to infection is the production of matrix metalloproteinases (MMPs), enzymes that directly degrade collagen, elastin, and other connective proteins. This is thought to allow immune cells to infiltrate hard-to-reach areas of the body by breaking down collagen and relaxing barriers to the immune response. Interestingly, there is a reciprocal link between MMP expression and inflammation: MMP is produced during the inflammatory response, but it also appears to regulate inflammation itself.
Other features typical of ME/CFS might also point to connective tissue degradation. Mast cell activation syndrome is highly co-morbid with both hEDS and ME/CFS, and when Mast cells are activated (“degranulate”), one of the key products produced is the enzyme tryptase, which is also known to both degrade connective tissue itself and may also result in downstream activation of MMPs that further weaken the tissue (although tryptase can also lead to enhanced expression of collagen in the form of fibroblasts, so this story may be more complicated). Mast cells in general are thought to play a key role in both degrading and remodeling connective tissue, which suggests that chronic activation of mast cells (a common feature of ME/CFS and long COVID) may be key to the persistence of the disease by either continually degrading already-damaged tissue (resulting in progressive deterioration), or instead inhibiting usual process of the healing after the virus is cleared.
We now have all the required conceptually machinery required to propose an actual model of post-viral ME/CFS onset and persistence.
The Model
So here is my “brainstem model of ME/CFS” (such as it is):
Acute infection with a virus or bacterium prompts widespread immune response that begins the process of degrading connective tissue throughout the body.
In some people, this process causes the physical integrity of key structures to fail. This may be because of a pre-existing weakness (as in the case of hEDS) or hyperactivation of the immune system.
This loss of integrity leads to pressure on the brainstem (especially the cNTS). This may be due the weight of the skull literally crushing the brainstem, or due to increased intracranial pressure caused by reduced venous outflow.
This structural damage silences or damages the cNTS and other key brainstem areas regulating autonomic and immune response.
Widespread hyperinflammation ensues, which prompts the expression of more MMPs, Mast cell activation, etc. which either continues to degrade connective tissue or keeps it from healing.
Long-term immune dysfunction allows dormant viruses to reactivate, exacerbating the whole wicked feedback loop.
The longer this loop persists, the more positive feedback can damage or derange other systems. For example, I haven’t touched much on point six (the ubiquitous reactivation of dormant viruses like EBV), but I included it at the end to illustrate how an initial insult to connective tissue integrity could ultimately lead to second order effects. EBV reactivation may not be the “root cause” in this model, but years of activated EBV in circulation will certainly trigger its own symptoms and problems (which may be manageable with antivirals). I suspect that much of the individual variability in ME/CFS presentation probably stems in part from these different second-order, positive feedback loops. Every person’s individual presentation is unique, and these differences probably reflect random, individual differences in pathway sensitivities, pre-existing risk factors, etc.
Relationship to the Itaconate shunt hypothesis
A nice feature of this hypothesis is that it potentially answers a standing question at the core of another popular model of ME/CFS: the itaconate shunt hypothesis (ISH). The ISH is a proposal by Dr. Rob Phair (with collaborators) to explain the chronic energy-limitations seen in ME/CFS patients. Health Rising has a good explanation of all the gory details, so I’ll give a surface-level explanation here, and those interested in the nitty gritty biochemistry can follow the above links.
Ordinarily, energy is produced in the mitochondria (the proverbial powerhouse of the cell) via the citric acid cycle: energy-rich fuels derived from carbohydrates or fatty acids go into the cycle, and out come useful products like NADH and ATP, which provide the energy required to run all of the cellular machinery. Ordinarily, all the mitochondria are healthy and happily chugging along, but his process can become disrupted when the innate immune system ramps up to start fighting infection. When this happens, the immune system begins producing an enzyme called cis-aconitate decarboxylase (CAD), which inserts itself into the citric acid cycle and effectively turns it off. This is the “itaconate shunt” in action.
This process is thought to be an adaptive response to infection: turning off cellular energy production means that invading pathogens can’t use it; since viruses need to leverage cellular metabolism to reproduce themselves, throttling energy production should also slow viral replication - giving the immune system time to get to work. So far, so good. The problem, though, emerges when the shunt doesn’t turn off. Ordinarily (the hypothesis goes), once the infection is cleared, the immune system quiets down and stops producing CAD, which in turn lets the citric acid cycle come back online. In people with ME/CFS and other forms of post-viral fatigue, however, it is possible that the itaconate shunt remains chronically on: cellular energy production simply never comes back online, and the body is forced to operate using other, less efficient forms of metabolism (such as the GABA shunt).
The ISH is compelling for a number of reasons, as it unifies many different features of ME/CFS: the tendency to appear after viral infection, disrupted energy production, etc, and suggests an explanation for the “poisoned” feeling typical of PEM: ammonia poisoning from compensatory amino-acid metabolism. However, there is currently not (as far as I can tell), an accepted explanation as to why the itaconate shunt remains on. I propose that, in some people, the shunt may remain on due to aberrant activation of the innate immune system, caused by disruption to the brainstem. The discovery of hyperinflammation following cNTS suppression might provide an answer. While Jin et al., didn’t look at production of the CAD enzyme at all, it seems possible that a consequence of this immune activation would be the initiation, and maintenance, of the itaconate shunt.
Relationship to the cell danger response hypothesis
The “cell danger response” (CDR) hypothesis is another theory about the etiology of ME/CFS (and a variety of other chronic illnesses. Proposed by Robert Naviaux, the CRD is a widely conserved set of metabolic and functional changes observed in cells when they are in danger (this danger can be toxic, viral, or mechanical threats). Naviaux has implicated the CRD in pretty much every chronic illness you can imagine, from post-infectious syndromes like ME/CFS and chronic Lyme disease, to neurodegenerative disorders like Alzheimers, to developmental differences like autism. Personally, I suspect he is getting out ahead of his skis a bit here, but that’s just my opinion. In the context of ME/CFS, Naviaux et al., propose that the CDR gets stuck in the “on” state, prompting a cascade of changes as the body’s cells begin to enter a hypometabolic state meant to protect them from acute, short-term damage. As with the ISH, the CDR hypothesis has a crucial question at its core: why does the CDR become stuck in the “on” position? What goes wrong that stops the cells from being able to relax? Once again, silencing the cNST might be the answer: the widespread, hyperinflammation observed by Jin et al., may result in the production of signals to the body that there is acute, biological danger at hand, triggering a widespread cell danger response transformation.
I personally remain somewhat agnostic to the CDR (I rather prefer to the ISH, of the two), but it would be very easy to test whether cNST silencing produces the kind of purinergic signaling thought to trigger the CDR.
Studies I’d like to see done
Any good model or hypothesis should make testable predictions; below are a set of eleven possible studies, ranging from human studies and clinical trials to basic research using animal models and post-mortem tissue analyses that could provide targeted tests of core features of this model. Any interested scientist with the funds, resources, and inclination should consider these to be freebies - I don’t care in the slightest about whether this blog post gets cited or anything like that, I just want to see the science done.
A trial testing whether cranio-cervical traction improves symptoms in ME/CFS. This might help us get a sense of what percentage of sufferers have mechanical brainstem issues as a root cause of their suffering.
A clinical trial of Diamox for ME/CFS - as with the traction study, this would help us understand how many people have IIH as a root cause.
I would like to see the Bragee study (which was published in 2020, just before long COVID became a concern) replicated with the largest possible patient size. The ballooning population of ME/CFS patients post-COVID should make it possible.
Comparison of connective tissue integrity in people with ME/CFS and compare it to healthy controls. This could be done post-mortem using tissue banks, or with tests typically used to diagnose hEDS.
More detailed studies of how Mast cell degranulation impacts connective tissue, with a focus on MCAS developed as a post-infectious sequelae.
Is MMP expression chronically higher in ME/CFS patients compared to healthy volunteers? If so, that might suggest ongoing damage to connective tissue that either causes progressive worsening of symptoms (as in the case of JB and JW) or inhibits healing.
Detailed animal studies of connective tissue integrity changes following acute infection with COVID-19, Epstein-Barr virus, and Borrelia burgdorferi.
Whether suppression of the cNTS triggers CAD expression (which would directly link brainstem dysfunction to the itaconate shunt hypothesis.
Whether suppression of the cNTS triggers MMP expression (which would substantiate the link between brainstem dysfunction and connective tissue degradation).
Whether suppression of the cNTS activate purinergic signaling, triggering a widespread CDR as part of the observed hyperinflammation.
Whether the CDR is involved in connective tissue degradation or remodeling?
Given a sufficiently large N, I think that any one of these studies could provide deep insights into the pathophysiology of ME/CFS, and in the case of the traction and Diamox studies, might also alleviate some suffering while at it.
All models are wrong, some are useful
I want to end by stressing that this is a model of how ME/CFS and long COVID might form, in some populations. This is almost certainly not going to be true of everyone, nor is it remotely likely to be the whole story, even in people for whom it is broadly accurate. It certainly will not explain every symptom in every person. That’s fine: there is a quote in my own field of complex systems science generally attributed to statistician George Bog:
“All models are wrong, but some are useful.”
When dealing with complex systems, like human biology or multi-system syndromes like ME/CFS, it is probably impossible to construct the One True Universal Model. There is too much noise, too many individual differences, and too many multi-part interactions between subsystems for any single scientist, doctor, or patient to hold in their minds simultaneously. That’s why we build models: simplified hypotheses that don’t capture the whole truth, but nevertheless let us focus our thinking and can provide insights into small subsets of the unmanageable “whole”. Models help us get a grip on complexity.
Models can also be useful for helping narrow down the space of possible interventions to test. Considering the huge number of possible drugs, supplements, interventions, and other possible therapeutic treatments, it is impractical to imagine running large-scale clinical trials on all of them, and ludicrous to imagine running clinical trials on all possible combinations. Given ten possible interventions, there are 115,975 possible combinations to try, which is obviously beyond the reach of even the most generous government funding program (and there are far more than ten possible interventions). A “throw-everything-at-the-wall-and-see-what-sticks” approach is a waste of resources, and more crucially, a waste of time - time that suffering patients don’t have to waste. By building these small models and testing specific hypotheses, we can engage in a process of iterative refinement, sequentially building, testing, and improving models, and hopefully helping some subset of patients improve at each step along the way.
If you are a clinician, scientist, or interested patient, and want to talk to me about the ideas presented here, you can email me at:
tfvarley at uvm dot edu



excellent
Wonderful. Nails down my own experience!